The global burden of renal pathology is undergoing a seismic shift, characterized by the rising incidence of Acute Kidney Injury (AKI) and Chronic Kidney Disease (CKD), particularly in developing economies like India. This report provides an exhaustive analysis of the clinical landscape of kidney failure, the pathophysiological mechanisms driving renal collapse, and the critical role of advanced medical technology in mitigating these outcomes. Central to this analysis is the evaluation of the Hemant Surgical Industries Ltd. (HSIL) “Nephrocure” ecosystem—a comprehensive portfolio of dialysis machines, water treatment systems, and consumables designed to address the specific epidemiological and infrastructural challenges of the Indian subcontinent. By synthesizing clinical data on tropical AKI, hemodynamic stability during renal replacement therapy (RRT), and the physics of high-flux hemodialysis, this document establishes a foundational understanding for nephrologists, hospital administrators, and healthcare policymakers. It posits that the successful management of kidney failure requires a symbiotic relationship between early clinical detection and the deployment of precision-engineered, cost-effective medical infrastructure.
Part I: The Epidemiology and Pathophysiology of Acute Kidney Injury
1.1 The Silent Epidemic: Defining Acute Kidney Injury
Acute Kidney Injury (AKI), historically termed acute renal failure, represents a sudden and often catastrophic decline in renal filtration function. Clinically, it is defined by a rapid increase in serum creatinine, a decrease in urine output (oliguria), or both, occurring within hours to days. Unlike the slow, inexorable progression of Chronic Kidney Disease (CKD), AKI is a precipitous event that destabilizes the body’s homeostatic balance, leading to the accumulation of nitrogenous waste products (azotemia) and lethal electrolyte disturbances.
The kidneys are metabolically active organs receiving approximately 20-25% of the cardiac output. This high perfusion requirement makes them uniquely susceptible to ischemic injury and toxic insults. When filtration collapses, the systemic consequences are immediate. Fluid retention leads to pulmonary edema and respiratory distress; hyperkalemia (elevated potassium) risks cardiac arrest; and uremic toxins impair neurological function, leading to encephalopathy. The reversibility of AKI is its defining clinical feature and its greatest challenge. With timely intervention, the renal parenchyma can regenerate; without it, the patient risks mortality or a transition to permanent End-Stage Renal Disease (ESRD).
1.2 The Indian Context: Tropical Fevers and Community-Acquired AKI
While AKI is a global challenge, its etiology in India presents a distinct epidemiological profile. Western literature often focuses on AKI in the context of elderly patients with multiple comorbidities or post-surgical complications. In contrast, Indian data reveals a high incidence of Community-Acquired AKI (CA-AKI) affecting younger, productive demographics. Recent multicenter prospective cohort studies indicate that tropical febrile illnesses are a predominant cause of CA-AKI in this region.
1.2.1 The Tropical Axis of Renal Injury
The monsoon season in India brings a spike in vector-borne diseases, which are potent triggers for AKI. The pathophysiology varies by pathogen but often involves a combination of hemodynamic collapse and direct tissue toxicity.
- Leptospirosis: Identified as the most nephrotoxic acute febrile illness in several Indian studies, leptospirosis causes AKI through direct tubular invasion by the spirochete and tubulointerstitial nephritis. It is frequently associated with severe outcomes requiring Renal Replacement Therapy (RRT).
- Dengue Hemorrhagic Fever: Dengue induces AKI primarily through hemodynamic instability. The massive capillary leak syndrome characteristic of severe dengue leads to intravascular volume depletion and prerenal failure. Furthermore, dengue-associated rhabdomyolysis can release myoglobin, which is directly toxic to renal tubules.
- Malaria: Plasmodium falciparum infection causes “blackwater fever,” where hemolysis leads to hemoglobinuria. The free hemoglobin obstructs renal tubules, while the parasitized red blood cells reduce microvascular flow in the renal medulla, causing ischemic necrosis.6
- Scrub Typhus: An increasingly recognized cause, this rickettsial infection leads to vasculitis and organ failure, including the kidneys.
The implication for healthcare providers is clear: in the Indian context, a patient presenting with fever and reduced urine output requires an immediate assessment for these tropical pathogens alongside standard renal function tests.
1.3 Etiological Classification: Prerenal, Intrinsic, and Postrenal
To understand the utility of dialysis interventions, one must first dissect the causes of AKI, traditionally categorized into three anatomical and physiological buckets.
1.3.1 Prerenal AKI: The Crisis of Perfusion
Prerenal AKI accounts for the majority of cases and is fundamentally a problem of blood flow. The kidney tissue is intact, but it is starved of the hydraulic pressure necessary for glomerular filtration.
- Volume Depletion: Severe dehydration from gastroenteritis (cholera, rotavirus), hemorrhage (trauma, postpartum bleeding), or excessive diuretic use reduces effective circulating volume.
- Decreased Cardiac Output: In heart failure or cardiogenic shock, the heart cannot pump sufficient blood to the kidneys. This “Cardiorenal Syndrome” creates a vicious cycle where fluid retention from failing kidneys places further strain on the failing heart.
- Systemic Vasodilation: Conditions like sepsis or anaphylaxis cause blood vessels to dilate widely. Although total blood volume may be normal, the pressure drops so low that the kidneys cannot filter. This is a critical mechanism in septic AKI, the leading cause of death in ICUs.
1.3.2 Intrinsic AKI: Structural Damage
When prerenal insults are prolonged, they evolve into intrinsic damage. The cells of the renal tubules, deprived of oxygen, undergo necrosis (Acute Tubular Necrosis or ATN).
- Ischemic ATN: The aftermath of prolonged shock or hypotension.
- Nephrotoxic ATN: Caused by exogenous toxins. In India, the unregulated use of Over-The-Counter (OTC) painkillers (NSAIDs) is a significant contributor. NSAIDs constrict the afferent arteriole, reducing blood flow to the glomerulus. When combined with ACE inhibitors (which dilate the efferent arteriole) and dehydration, the filtration pressure collapses—a “triple whammy” for the kidney. Other toxins include aminoglycoside antibiotics, radiographic contrast media, and heavy metals.
- Glomerulonephritis: Autoimmune conditions like Lupus (SLE) or post-infectious glomerulonephritis cause direct inflammation of the filtration units.
1.3.3 Postrenal AKI: The Blockade
This is the least common but most treatable form. It results from obstruction of urine flow.
- Causes: Kidney stones (renal calculi), Benign Prostatic Hyperplasia (BPH) in older men, or malignancies (cervical, prostate, bladder cancers) compressing the ureters.
- Mechanism: The obstruction causes back-pressure (hydronephrosis) which opposes the filtration pressure. Once the pressure in Bowman’s capsule equals the glomerular capillary pressure, filtration stops.
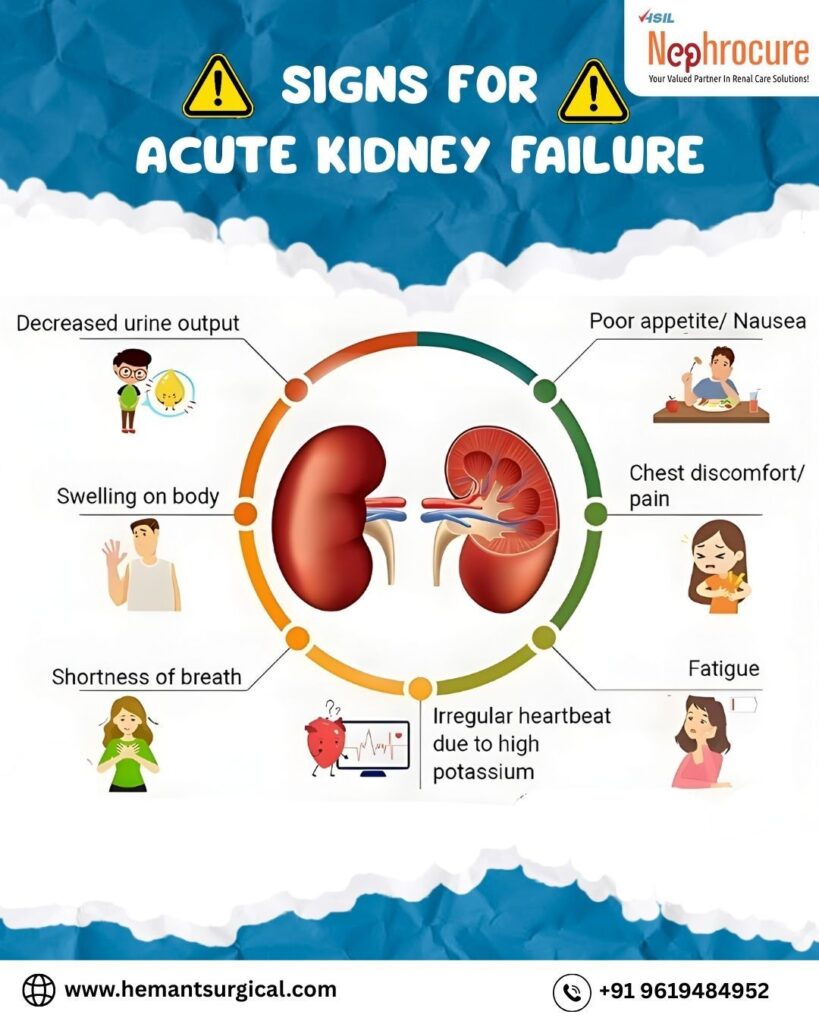
1.4 Clinical Manifestations and the Window of Opportunity
The symptoms of AKI are often subtle until the damage is severe. The “silent” nature of early AKI underscores the need for vigilant monitoring of urine output and biochemistry in hospitalized patients.
| System | Symptom/Sign | Pathophysiology |
| Renal | Oliguria (<400ml/day) or Anuria | Reduced GFR; Tubular blockage. |
| Cardiovascular | Hypertension, Edema, Arrhythmia | Fluid overload; Hyperkalemia affecting cardiac conduction. |
| Respiratory | Dyspnea, Crackles, Kussmaul breathing | Pulmonary edema; Metabolic acidosis (respiratory compensation). |
| Neurological | Confusion, Lethargy, Seizures | Uremic encephalopathy; Cerebral edema from hyponatremia. |
| Gastrointestinal | Nausea, Vomiting, Anorexia | Uremic toxins affecting the GI mucosa (uremic gastritis). |
Table 1: Systemic Manifestations of Acute Kidney Injury.
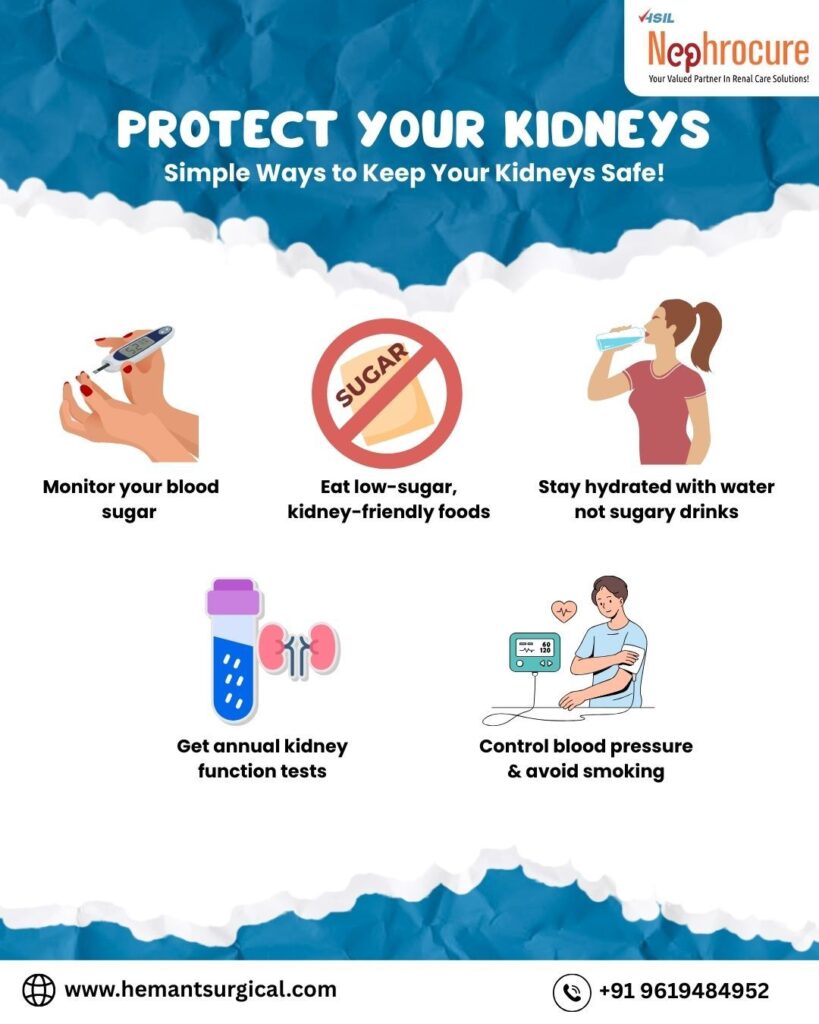
The KDOQI and KDIGO guidelines emphasize that early recognition is the single most significant factor in preventing mortality. The “0 by 25” initiative by the International Society of Nephrology aims to eliminate preventable deaths from AKI by 2025, a goal that requires massive improvements in diagnostic capability and treatment accessibility in regions like India.
Part II: The Technological Imperative – Hemodialysis in AKI
2.1 The Role of Renal Replacement Therapy (RRT)
When conservative management (fluids, diuretics, dietary restriction) fails to correct the metabolic abnormalities of AKI, Renal Replacement Therapy (RRT) becomes a life-saving necessity. The decision to initiate dialysis is guided by the AEIOU criteria: Acidosis, Electrolyte abnormalities (refractory hyperkalemia), Intoxication, Overload (fluid), and Uremia.
In the context of AKI, dialysis is not merely a “cleaning” process; it is a critical support system that buys time for the kidneys to heal. However, the procedure itself carries risks, primarily hemodynamic instability. A patient with septic shock or severe cardiac compromise may not tolerate the rapid fluid shifts of conventional hemodialysis. This necessitates the use of advanced dialysis machines capable of precise volumetric control and sequential therapy modes.
2.2 Hemodynamic Stability: The Holy Grail of Acute Dialysis
Intradialytic hypotension (IDH) is a frequent complication in AKI patients. A drop in blood pressure during dialysis can cause further ischemic injury to the already recovering kidneys, potentially converting reversible AKI into permanent ESRD.
- Sequential Dialysis (ISO-UF): This modality separates the removal of fluid (ultrafiltration) from the removal of solutes (diffusion). By removing fluid without the osmotic shift caused by toxin removal, or vice versa, the vascular space has time to refill from the interstitium, maintaining blood pressure.16
- Sodium Profiling: Modern machines allow the programming of a sodium gradient in the dialysate. Starting with a higher sodium concentration helps draw fluid into the vascular space, supporting blood pressure during the early phase of rapid fluid removal.18
- Temperature Control: Cooling the dialysate induces vasoconstriction, which helps maintain blood pressure during treatment.16
These sophisticated maneuvers require equipment that goes beyond basic filtration. They demand intelligent, computer-controlled systems capable of real-time monitoring and feedback—a technological gap that HSIL aims to bridge with its SWS series.
Part III: The HSIL Nephrocure Ecosystem – Engineering Clinical Excellence
3.1 A Legacy of Medical Innovation: Hemant Surgical Industries Ltd.
The story of advanced renal care in India is incomplete without acknowledging the infrastructure providers. Hemant Surgical Industries Ltd. (HSIL) stands as a paradigm of the evolution of the Indian medical device sector. Incorporated in 1985, HSIL began its journey as an importer, most notably establishing an exclusive partnership with JMS Co. Ltd. (Japan). This 30-year collaboration introduced Japanese standards of precision manufacturing—known for rigor and zero-defect philosophies—into the Indian market.
HSIL has recently pivoted from pure distribution to manufacturing, embodying the “Make in India” ethos. With manufacturing units in Achhad (Gujarat) and Atgaon/Bhiwandi (Maharashtra), and a new facility announced in Shambhaji Nagar in 2025, HSIL has localized the production of critical dialysis components. This localization is strategic: it insulates the Indian healthcare market from global supply chain shocks (a lesson learned during the COVID-19 pandemic) and reduces the cost of care, making dialysis accessible to the lower socio-economic strata most vulnerable to CA-AKI.
3.2 The Core Engine: HSIL Nephrocure SWS 4000A Dialysis Machine
The centerpiece of HSIL’s renal portfolio is the SWS 4000A Hemodialysis Machine. An analysis of its specifications reveals a design philosophy strictly aligned with the clinical needs of AKI management described in Part II.
3.2.1 Technical Specifications and Architecture
The SWS 4000A is a compact, mobile unit designed for the rigorous environment of ICUs and high-volume dialysis centers.
- Dimensions & Weight: Standing at 145cm x 46cm x 50cm and weighing approximately 60kg, it is engineered for stability yet is mobile enough to be moved to a patient’s bedside in an ICU setting.
- Electrical Robustness: Operating on 220V/50Hz (or 240V/60Hz), the machine includes a critical Battery Backup system capable of running the extracorporeal circuit for at least 40 minutes. In the Indian context, where power fluctuations are common, this feature is non-negotiable. It allows for the safe return of blood to the patient in the event of a total power failure, preventing blood loss and clotting within the circuit.
3.2.2 Advanced Therapy Modes for Critical Care
The SWS 4000A distinguishes itself with software and hardware tailored for hemodynamically unstable patients.
- Sequential Dialysis (Dialysis ↔ IUF): The machine supports isolated ultrafiltration (IUF) and sequential therapies. This allows the nephrologist to tailor the treatment: removing fluid first to reduce lung edema, then performing diffusion to clear toxins, or strictly controlling the rate to prevent hypotension.
- Profiling Capabilities: It offers customizable Sodium and Ultrafiltration profiles. This means the rate of fluid removal can be high initially and taper off, or match the patient’s physiological refill rate, significantly reducing the incidence of cramps and hypotension.
- Kt/V Calculation: The machine provides real-time calculation of Kt/V (a measure of dialysis adequacy). This ensures that even in acute settings, the patient receives a therapeutic dose of dialysis, which is correlated with better survival outcomes.
3.2.3 Safety Systems: The Japanese Influence
Reflecting the quality standards of its partners, the SWS 4000A incorporates redundant safety mechanisms.
- Air Detection: An ultrasonic air bubble detector on the venous line automatically clamps the tube if air is detected, preventing fatal air embolisms. The machine also features an automatic liquid level adjustment in the venous chamber, reducing the risk of air entering the system—a feature often found only in high-end European models.
- Blood Leak Detection: Optical sensors monitor the dialysate for traces of blood, indicating a membrane rupture. This is crucial for AKI patients who may be on anticoagulants and cannot afford blood loss.
- Component Sourcing: The reliability is bolstered by global sourcing of critical components: Pressure sensors from the USA, Conductivity meters from Germany, and Electronic boards from Japan. This hybrid engineering ensures durability.
3.3 The Physics of Filtration: Nephrocure High Flux Dialyzers
If the machine is the heart of the system, the dialyzer is the kidney. HSIL’s Nephrocure High Flux Dialyzers represent the forefront of membrane technology.
3.3.1 High Flux vs. Low Flux: A Clinical Distinction
Dialyzers are classified by their “flux,” or permeability.
- Low Flux: These membranes have smaller pores and primarily remove small molecules like urea and creatinine via diffusion. They are adequate for basic survival but fail to remove larger toxins.
- High Flux: These membranes have larger pores and higher hydraulic permeability. They allow for the removal of “Middle Molecules” (molecular weight 500–60,000 Daltons), such as Beta-2 Microglobulin and various inflammatory cytokines involved in sepsis.
Clinical Relevance for AKI: In septic AKI, the blood is laden with inflammatory mediators. High flux dialysis can help clear these cytokines (convective clearance), potentially dampening the systemic inflammatory response. The Nephrocure dialyzers (available in 1.4, 1.6, 1.8, and 2.0 m² surface areas) facilitate this enhanced clearance while retaining essential proteins like albumin.
3.3.2 Biocompatibility and Sterilization
The Nephrocure dialyzers utilize synthetic membranes (typically Polysulfone-based) which demonstrate superior biocompatibility compared to older cellulose membranes. This minimizes complement activation and neutropenia, reducing the oxidative stress on the patient. Furthermore, HSIL emphasizes the use of sterile, single-use dialyzers where possible to prevent cross-infection, although they also provide reprocessing equipment for cost-constrained settings.
Part IV: Water Treatment – The Forgotten Drug
In hemodialysis, water is not just a utility; it is a “drug.” A patient is exposed to 120-150 liters of water across a semi-permeable membrane during each session. Any contaminant in the water—bacteria, endotoxins, heavy metals—can cross into the blood, causing pyrogenic reactions, chronic inflammation, and “micro-inflammation,” which contributes to cardiovascular disease and anemia.
4.1 HSIL Nephrocure Reverse Osmosis (RO) Plants
HSIL manufactures Stainless Steel (SS) RO Plants with capacities ranging from 100 to 3000 Liters Per Hour (LPH). The choice of stainless steel (SS304/316) over PVC for piping and skids is critical. Stainless steel minimizes biofilm formation and allows for heat sanitization, ensuring the highest hygiene standards.
4.2 The Superiority of Double Pass RO Technology
For optimal patient safety, especially in AKI cases where the immune system is compromised, HSIL advocates for Double Pass RO Systems.
| Feature | Single Pass RO | Double Pass RO | Clinical Implication |
| Process | Feed water passes through membranes once. | Product water from Pass 1 becomes feed for Pass 2. | Double Pass provides redundancy; if Pass 1 fails, Pass 2 protects the patient. |
| Conductivity | Typically < 20 µS/cm. | Typically < 5 µS/cm. | Lower conductivity implies fewer dissolved ions and contaminants. |
| Endotoxin Rejection | 99% | > 99.9% | Critical for producing “Ultrapure Dialysate.” |
| Application | Standard Chronic Dialysis. | High Flux Dialysis / HDF / Acute Care. | Essential for preventing pyrogenic reactions in ICU settings. |
Table 2: Comparison of Single vs. Double Pass RO Systems.
By utilizing Double Pass technology, HSIL ensures that the water quality meets or exceeds AAMI/ISO standards for ultrapure water. This ultrapure water is essential when using High Flux dialyzers; because high flux membranes are so permeable, “back-filtration” of dialysate into the blood can occur. If the water is not sterile, this back-filtration injects endotoxins directly into the patient. The combination of Nephrocure High Flux Dialyzers and Double Pass RO plants eliminates this risk.
4.3 Endotoxin Filters and Disinfection
To further guarantee sterility, HSIL integrates Endotoxin Filters (ultrafilters) into the fluid path. These filters remove bacterial fragments (LPS) that might survive RO treatment. The system is supported by Nephrocure Cold Sterilant and heat disinfection protocols, ensuring that the “dead legs” (stagnant water zones) in the piping are minimized and regularly sanitized.
Part V: The Consumable Circuit and Vascular Access
The efficacy of the dialysis machine is entirely dependent on the integrity of the extracorporeal circuit—the tubing and needles that connect the patient to the device.
5.1 Vascular Access: The Lifeline
For AKI patients, immediate access is usually achieved via a central venous catheter. HSIL supplies Nephrocure Double Lumen Catheters designed for jugular, subclavian, or femoral insertion. These catheters must be rigid enough to insert but flexible enough to not damage the vessel wall.
For chronic patients, the AV Fistula is the gold standard. HSIL’s AV Fistula Needles (15G, 16G, 17G) feature an ultra-thin wall and a back-eye design. The back-eye (a small hole on the back of the needle bevel) ensures smooth blood flow even if the needle tip is resting against the vessel wall, preventing turbulence and hemolysis.
5.2 Blood Tubing Sets (BTS)
The Nephrocure Blood Tubing Set acts as the extension of the patient’s vascular system. It is manufactured from medical-grade PVC that is biocompatible and resistant to kinking. Crucially, the “pump segment” of the tubing is engineered to withstand the mechanical stress of the peristaltic pump rollers for 4+ hours without spallation (shedding of micro-plastic particles into the blood).
5.3 Pharmaceutical Support
HSIL provides the pharmacological agents necessary to run the dialysis session:
- Heparin Sodium: Essential for anticoagulation to prevent the blood from clotting in the circuit.
- Iron Sucrose: To manage the anemia of chronic disease.
- Dialysis Concentrates: The acidic (Part A) and bicarbonate (Part B) solutions that determine the electrolyte composition of the dialysis fluid. HSIL’s provision of Bicarbonate Mixers allows centers to mix their own solution from powder, significantly reducing logistics costs compared to transporting liquid canisters.
Part VI: Infrastructure Solutions – The Turnkey Model
The challenge in India is not just the lack of machines, but the lack of functional dialysis centers, particularly in Tier 2 and Tier 3 cities. HSIL addresses this through its Turnkey Dialysis Setup service.
6.1 Building Capacity from Scratch
HSIL has successfully installed over 4000 dialysis machines across Asia and Africa, leveraging a model that goes beyond equipment supply. A turnkey project involves:
- Site Feasibility and Design: Planning the layout to ensure infection control (separating Hepatitis B/C positive machines), optimizing water loop routing, and ensuring electrical safety.
- Equipment Provisioning: Supplying the full suite: SWS 4000A machines, RO plants, motorized dialysis chairs (for patient comfort during 4-hour sessions), and defibrillators/monitors for emergency crash carts.
- Technical Training: A dedicated team of 45+ biomedical engineers provides installation and ongoing training for hospital staff. This is critical in rural areas where technical expertise is scarce. The training covers machine operation, RO plant maintenance (backwashing, membrane cleaning), and emergency troubleshooting.
6.2 Economic Viability and Public Health
The cost of dialysis is a major barrier to access. By manufacturing key components in India and offering refurbished options (like the Fresenius 4008S), HSIL lowers the capital expenditure (CAPEX) for new centers. This reduction in CAPEX allows hospitals to offer dialysis at lower price points, aligning with government schemes like the Pradhan Mantri National Dialysis Program (PMNDP). Feasibility studies in districts like Bijapur have shown that such PPP (Public-Private Partnership) models are the only viable way to provide care to the 37% of the population living below the poverty line.
For inquiries about our products, please contact:
Mail us: sales@hemantsurgical.com
Contact us: +91 961 948 4952
Visit us at: https://nephrocure.in | https://hemantsurgical.com
